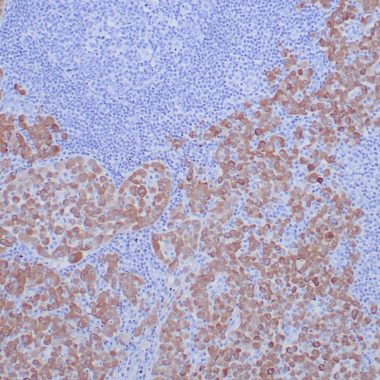
ROS1, an orphan receptor tyrosine kinase of the insulin receptor family, was initially identified as a homolog of v-ros from the UR2 sarcoma virus. ROS1 consists of a large extracellular domain composed of six fibronectin repeats, a transmembrane domain, and an intracellular kinase domain. While the function of ROS1 is undefined, it has been shown to play an essential role in the differentiation of epididymal epithelium. The first oncogenic fusion of ROS1, FIG-ROS1, was initially identified by research studies in glioblastoma. Investigators have found additional oncogenic ROS1 fusion proteins in NSCLC (at a frequency of ~1.6%), where the ROS1 kinase domain is fused to the amino-terminal region of a number of different proteins, including CD74 and SLC34A2.