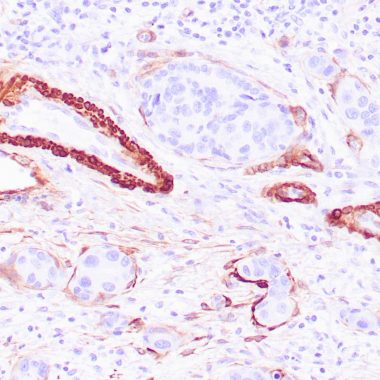
Multiple isoelectric variants of calponin have been identified, however only two molecular weight isoforms exist; a 34kDa form and a 29kDa form. Expression of the 29kDa form, I-calponin, is primarily restricted to muscle of the urogenital tract, whereas the higher molecular weight variant has been demonstrated in vascular and visceral smooth muscle. In Western blotting, this MAb reacts with only the 34kDa form of calponin in extracts of human aortic medial smooth muscle and is unreactive with fibroblast extracts of cultivated human foreskin. Calponin is a calmodulin, F-actin and tropomyosin binding protein, which is thought to be involved in the regulation of smooth muscle contraction. Calponin expression is restricted to smooth muscle cells and has been shown to be a marker of the differentiated (contractile) phenotype of developing smooth muscle.